 With options for efficient target protein elution and tolerance of a range of feed conductivity, this technique often allows easy transition between chromatography unit operations, thus eliminating the need for dilution and extensive feed manipulation. Mixed-mode chromatography offers additional advantages for protein separation and purification, according to Dr. They were then refined and converted to a step elution protocol for larger scale production. Each of these methods was tested for yield and purity of target protein. He described an approach based on creating different types of buffer gradients on a traditional chromatographic column-pH, conductivity, or elution with additives. In the second purification method development strategy, Dr. 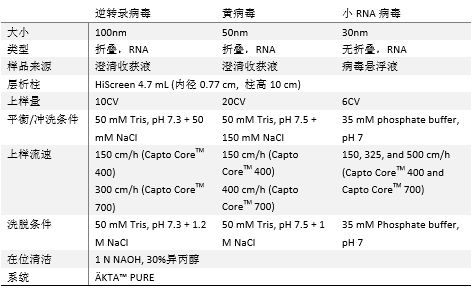
Purification conditions obtained from these DoE screening tests were experimentally confirmed as being effective for host cell protein and double-stranded DNA removal, and were suitable for use in production scaleup. In all of these experiments spin columns containing 50 µL of Nuvia cPrime media were used. The “sweet spots” for optimal yield and purity of lysozyme were identified using a set of 11 experiments that covered the pH range of 4–8 and NaCl concentration range of 10–400 mM for binding and 10–1,000 mM for elution. In the first example, she investigated the effect of buffer salt concentration and pH on the purity and yield of final product via a design of experiment (DoE) approach. He presented two different purification method development schemes and associated optimization strategies. Potential interaction modes for protein binding include weak cationic exchange, hydrophobic interaction, and hydrogen bonding. Nuvia cPrime is composed of a functional ligand on a base matrix of macroporous hydrophilic polymers. He cites the main advantages of Nuvia cPrime as high selectivity, tolerance of load conductivity, and gentle purification conditions. Xuemei He, Ph.D., senior staff scientist, process chromatography division, Bio-Rad Laboratories, described the use of Nuvia™ cPrime™, a hydrophobic cation exchange media, for mixed-mode chromatography for the purification of a variety of recombinant proteins expressed by prokaryotic or mammalian cell lines. This technology is increasingly being applied to other challenging nonantibody therapeutic proteins for which large-scale downstream separation and purification protocols often introduce unwanted process bottlenecks and cost barriers. Conclusion Comparing with Sepharose 6FF, Capto Core 400 removes residual Vero cell HCP and DNA more effectively, with more highly optimized process parameters, resulting in increased sPV purification process efficiency and lowered time and cost.Mixed-mode chromatography has proven to be a valuable alternative to conventional ion-exchange (IEX) or hydrophobic-interaction chromatography for purifying monoclonal antibodies primarily due to the technique’s high selectivity. The specific values for Capto Core 400 and Sepharose 6FF process parameter ratios were: medium volume 1/20, purification time 1/25, application volume of buffer 1/12, process linear velocity 10, loading quantity of sample per litre medium 20, maximum withsatand pressure 2. 
But HCP residue ( t values were 3.15, 3.23 and 3.54, respectively) and cell DNA residue removal rates (t values were 3.41, 3.25 and 3.62, respectively) of the former were all higher than the latter, all with statistically significant differences (all P values<0.05). Results D antigen recovery rates of types Ⅰ, Ⅱ, Ⅲ sPV purified by Capto Core 400 and Sepharose 6FF had no statistically significant difference ( t values were 1.09, 1.08, and 1.02, respectively, all P values>0.05). D antigen recovery rates, as well as HCP and DNA removal rates were caculated. D antigen contents, HCP and DNA residues in purified sPV liquids were detected. 
Methods Types Ⅰ, Ⅱ, Ⅲ sPV concentrates were purified using Capto Core 400 and Sepharose 6FF media, respectively. 
Abstract: Objective To purify Sabin strain poliovirus (sPV) using new multimodal chromatography medium Capto Core 400 and traditional medium Sepharose 6FF to remove residual host cell protein (HCP) and DNA, comparing purification results and process parameters. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
